a) What is Melanoma?
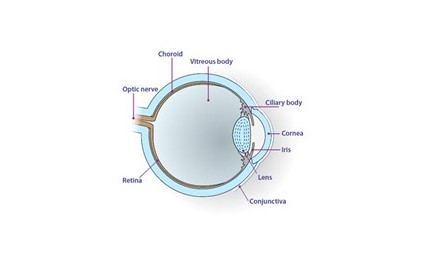
Eye melanoma is cancer of the eye. This is the most common type of eye cancer in adults. Most patients are between 55 and 65 years old when diagnosed. It affects up to 50 people a year in Scotland (between 500 and 600 people in the UK). Choroidal melanoma is the most common type of uveal melanoma, followed by the ciliary body, then iris. Uveal melanoma can spread to other parts of the body; some patients have spread, some don’t. The most common place for melanoma to spread to is the liver. This can happen many years after diagnosis. Unfortunately treating the tumour does not guarantee spread will not happen.
b) What are the symptoms of eye melanoma?
Eye melanoma may have no symptoms and may be picked up on routine examination by your optician. Some patients, however, may report:
• Decreased vision
• Flashing lights
• Shadow in the vision
Choidal, ciliary body and iris melanomas may have different symptoms.
i) Choroidal melanoma:
Patients may experience a shadow in the peripheral vision or poor central vision. As the melanoma grows it may cause a retinal detachment. This may cause floaters, flashing lights or a curtain over part of the vision.
ii) Ciliary body melanoma:
As the ciliary body is behind the iris, small tumours may be not be visible at first. They can, however, grow and cause a shadow in the vision. Clouding of the lens, known as cataract, can develop resulting in blurred vision. Floaters can be caused if the tumour bleeds into the back of the eye. Glaucoma (raised pressure in the eye) may occur causing poor vision and eye pain.
iii) Iris melanoma
Iris melanomas may appear as a growing pigmented area on the coloured part of the eye. This can change the shape of the pupil or grow in front of the pupil causing the vision to go down. Like Ciliary body melanoma, glaucoma (raised pressure in the eye) may occur causing poor vision and eye pain.
c) What are the risks of getting melanoma?
The following features may increase your risk of developing ocular melanoma:
• Pale skin
• Red or blond hair
• Blue eyes
• Over the age of 50
• Large number of moles or freckles
d) Will I need any tests?
Certain tests help confirm the diagnosis and check how the tumour is behaving. Tests may include:
• Photograph
• Ultrasound scan
• Optical Coherence Tomography (OCT)
All patients have an ultrasound of their liver every year to make sure the melanoma has not spread or started to grow here. This is usually organised at your local hospital.
e) Will I need a biopsy?
A biopsy may be offered to confirm the type of tumour and likelihood of spread. This, however, has risks and may not provide enough tissue to give a definite answer. If we do decide to biopsy, options will be discussed in clinic.
f) What is the treatment of eye melanoma?
In treating eye melanoma, our aim is remove or destroy the tumour and keep as much normal vision as possible. Unfortunately, removing the tumour does not guarantee spread will not happen later in life. Treatments may include:
- Plaque radiotherapy
- Proton beam radiotherapy
- Laser (Transpupillary thermotherapy)
- Photodynamic Therapy (PDT)
- Removal of eye (enucleation)
- Removal of tumour (local resection)
g) Is treatment always needed?
Very rarely we may choose not to treat. If the patient is frail, elderly, or has other serious medical illnesses we may decide to watch instead. Every patient is different. After discussion in clinic we will arrive at the right and best treatment option for you.
h) How likely is the melanoma to spread?
Risk of melanoma spreading (metastasising) to other parts of the body depends on the three factors outlined below.
i) Clinical tumour stage
Clinical tumour stage is based on how the tumour looks when examined in the clinic and the size of the tumour. The larger the tumour, the longer it is likely to have been there and the more likely it has had a chance to spread elsewhere in the body. If the tumour has grown quickly in size or grown through the wall of the eye, again this increases the risk of tumour spreading.
ii) Histology
This refers to how the tumour cells look under the microscope. Tumour tissue from biopsy or after the eye is removed is sent to the laboratory and examined by our pathologist. If the tumour cells appear large and round (epithelioid) then there is high chance of tumour spreading. If the cells appear long and narrow (spindle) then chance of spread is far less.
iii) Genetic typing
This refers to the arrangement of DNA in our cells (the basic building blocks for life). Our cell behaviour is controlled by DNA, which is stored in our genes. These genes are found in chromosomes, which are thread like structures found in every cell of our body. We have a total of 23 pairs of chromosomes. Each chromosome has long arms (coded by the letter q) and short arms (coded by the letter p). Tumour tissue is sent to the genetics laboratory where it is analysed. If a single chromosome 3 is missing from melanoma cells, this is called “monosomy 3”. This means the melanoma is very likely to spread. If chromosome 8q has gained a longer arm, again, this increases the chance of the melanoma spreading. If both “monosomy 3” and 8q gain are present then this there is an even stronger chance of tumour spread. If, however, there is a gain in chromosome 6p, this is protective and decreases the likelihood of metastasis.
i) Will I need any tests to check for spread?
We will perform an ultrasound scan (USS) of the liver every 6 months to check for spread of the melanoma. If we suspect there is spread on USS, we will organise an MRI scan of the liver to look at this in closer detail.
Some patients find having an USS every 6 months too stressful and worrisome. If this is the case we can perform the USS every 12 months instead.
j) What happens if the eye melanoma spreads to different parts of the body?
This can be scary and upsetting. If spread of disease has been picked up we involve other health professionals at our MDT (multidisciplinary team) meeting. Our clinical team consists of a radiologist, pathologist, and a medical oncologist. Collectively we will decide on the right tests and treatments for you as an individual. Accepting and coming to terms with the diagnosis can be challenging. For this reason, we have specialist ophthalmic nursing staff in the clinic who are here to council and help you through this difficult time.